 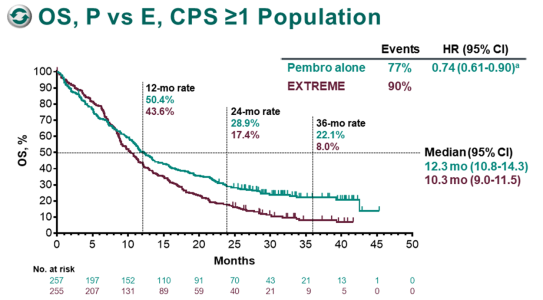 
The appearance of one or more new lesions was also considered PD. In addition to the relative increase of 20%, the sum had to demonstrate an absolute increase of ≥5 mm. Per RECIST 1.1, PD was defined as ≥20% increase in the sum of diameters of target lesions. PFS was defined as the time from randomization to the first documented progressive disease (PD) per RECIST 1.1 based on BICR, or death due to any cause, whichever occurred first. 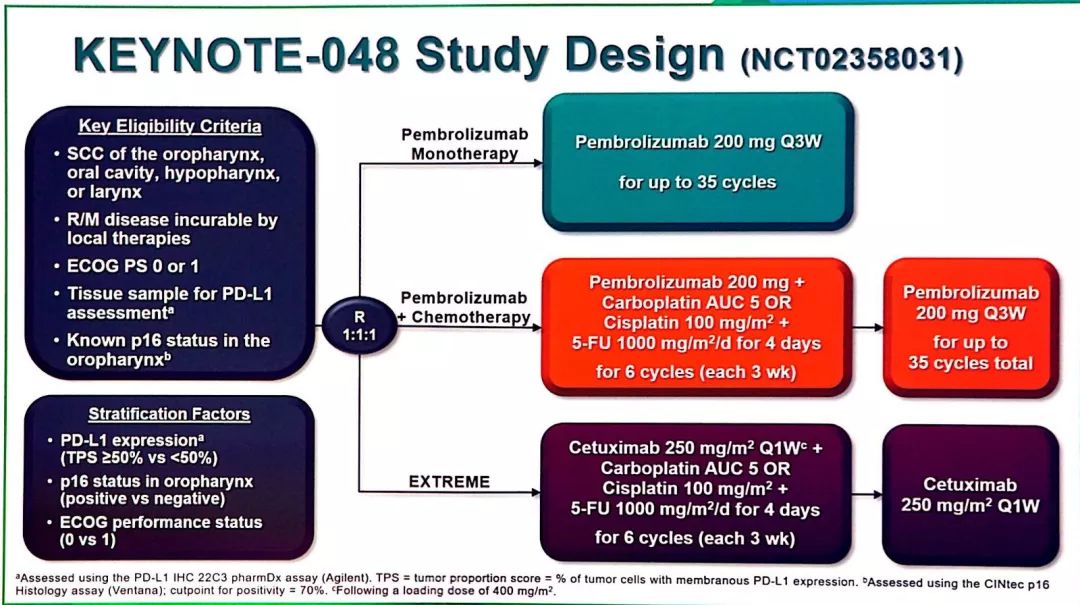
Recurrent Head and Neck Cancer Metastatic Head and Neck Cancerīiological: Pembrolizumab Drug: Cisplatin Drug: Carboplatin Drug: 5-FU Biological: Cetuximab The overall primary study hypotheses are as follows in all participants and in participants with Programmed Cell Death Ligand 1 (PD-L1) positive expression defined by Combined Positive Score (CPS) ≥1 and CPS ≥20: 1) pembrolizumab monotherapy prolongs progression free survival (PFS) per Response Evaluation Criteria in Solid Tumors Version 1.1 (RECIST 1.1) assessed by Blinded Independent Central Review (BICR) and prolongs overall survival (OS) compared to standard treatment, and 2) pembrolizumab combination with chemotherapy prolongs PFS per RECIST 1.1 assessed by BICR and prolongs OS compared to standard treatment. Participants with recurrent or metastatic (R/M) squamous cell cancer of the head and neck (HNSCC) will be randomly assigned to receive pembrolizumab monotherapy, pembrolizumab plus chemotherapy with a platinum-based drug (cisplatin or carboplatin) and 5-Fluorouracil (5-FU), or cetuximab plus a platinum-based drug (cisplatin or carboplatin) and 5-FU. Why Should I Register and Submit Results?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
